 As a result of its ion selectivity, an electrochemical membrane potential of about 180 mV builds up across the inner mitochondrial membrane. These can only get across with the aid of specific membrane transport proteins, each of which is selective for a particular ion or molecule. By contrast, the inner membrane is a tight diffusion barrier to all ions and molecules. Because of its porosity, there is no membrane potential across the outer membrane. Any larger molecules, especially proteins, have to be imported by special translocases. The outer membrane is porous and freely traversed by ions and small, uncharged molecules through pore-forming membrane proteins (porins), such as the voltage-dependent anion channel VDAC. In humans, only 13 mitochondrial proteins are organelle-encoded, all of them central, hydrophobic subunits of respiratory chain complexes or of the ATP synthase.Īs ubiquitous, semi-autonomous cellular organelles, mitochondria are separated from the cytoplasm by the outer and inner mitochondrial membrane (Fig. Today, the vast majority of mitochondrial proteins are produced in the cytoplasm and imported into the organelle by an elaborate set of protein translocases. In the course of evolution they have transferred up to 99 % of their genes to the nucleus. Nevertheless, mitochondria make surprisingly little use of their specialized protein production machinery. Very recently, the first high-resolution structure of a mitochondrial ribosome, determined by single-particle electron cryomicroscopy (cryo-EM), has revealed a fascinating patchwork of similarities to and differences from bacterial ribosomes. They have their own protein translation machinery, complete with ribosomes, tRNAs and associated protein factors that more or less resemble those of their bacterial ancestors. Mitochondria have their own genetic system, which uses a distinct DNA code that differs both from that of their bacterial ancestors and their eukaryotic hosts. The most striking evidence of this evolutionary relationship is the close homology of bacterial and mitochondrial respiratory chain complexes. In many respects, mitochondria resemble α-proteobacteria, from which they are thought to have originated by endocytosis some 1.6 billion years ago. Moreover, mitochondria are deeply implicated in apoptosis and ageing. Mitochondrial dysfunction is the cause of severe, often maternally inherited diseases. Not surprisingly, mitochondria play a fundamental role in human health. They also act in calcium signaling, stress responses and generally as cellular signaling hubs. Apart from cellular respiration and ATP synthesis, mitochondria have numerous other essential functions, including the production of NADH and GTP in the citric acid cycle, the biosynthesis of amino acids, heme groups and iron-sulfur clusters or the synthesis of phospholipids for membrane biogenesis. These are the products of ATP hydrolysis at the sites where energy is needed in the cell. ATP is generated by the mitochondrial ATP synthase from ADP and phosphate ions. ATP is produced on a massive scale in the human body, amounting to 50 kg per day in a healthy adult, but considerably more in a long-distance runner. These functions include force generation (for example, in muscle contraction and cell division), the biosynthesis, folding and degradation of proteins, and the generation and maintenance of membrane potentials. 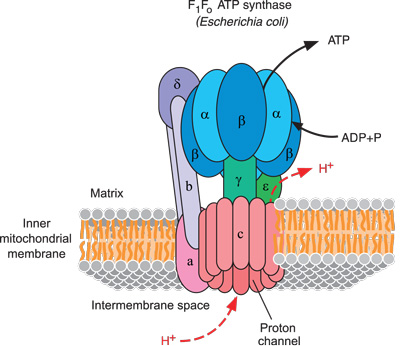
In all eukaryotes that do not depend on photosynthesis, the mitochondria are the main source of adenosine triphosphate (ATP), the energy-rich compound that drives fundamental cell functions. 
Mitochondria are the powerhouses of the cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
